Prof. Ester Segal: Multifunctional Nanoparticles
Prof. Segal from the Faculty of Biotechnology & Food Engineering, heads a multidisciplinary research team investigating the interface between materials science and biotechnology. The main research at the laboratory focuses on the synthesis and characterization of nanomaterials and their application to the development of biological sensors and drug delivery systems.
In an article she recently published in the prestigious journal Nature Communications, she reported that silicon carriers for the local delivery of anticancer drugs degrade differently when they reach the diseased environment, which can affect clinical outcomes. The study, conducted jointly with Prof. Natalie Artzi from the Massachusetts Institute of Technology (MIT) and the Harvard Medical School, sheds light on this degradation process, opening the way for improved tumor treatments.
“In this study we have shown for the first time that biomaterials in general, and nanostructured porous silicon in particular, behave differently when they are injected (or implanted) at the tumor microenvironment. Over the last few years, we successfully engineered silicon to be used as a carrier of anticancer drugs that releases its contents in a controlled manner, and now we are focusing on the degradation mechanism of the silicon at the diseased tissue,” explains Prof. Segal.
Nanostructured Porous Silicon is the common name for a family of silicon-based materials containing nano-scale holes. This material is today seen as a promising drug delivery vehicle, mainly due to its unique characteristics: a large surface area (geared for drug unloading), biocompatbility, and bio-degradability in a safe and non-toxic manner. .....read more
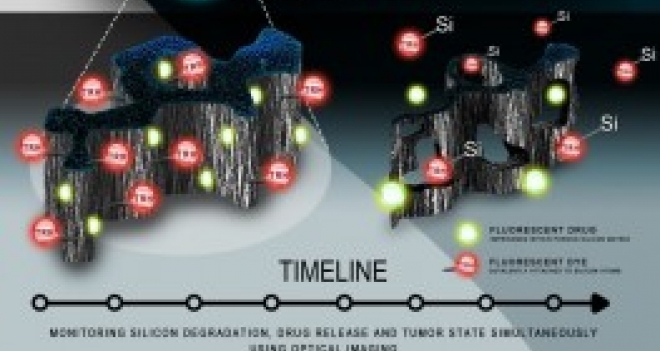
Mechanism of erosion of nanostructured porous silicon drug carriers in neoplastic tissues
The image illustrates porous silicon (PSi) particles used as a platform for the delivery of anticancer drugs (highlighted in green on the diagram) and its release at the targeted breast cancer tumors. The degradation of the PSi at the tumor microenvironment was investigated using novel imaging methods. The researchers tracked the decomposition of the material on the diseased tissue and uncovered its degradation mechanism, which triggers the release of the drugs trapped within the porous medium.